Lidocaine May Be Able to Kill Certain Cancer Cells by Activating Bitter Taste Receptors
Preclinical study explains the previously observed anti-cancer effect of the common anesthetic drug
(Penn Medicine News) — Lidocaine—often used as numbing agent for outpatient medical procedures—activates certain bitter taste receptors through two unique mechanisms that result in cancer cell death, according to researchers from the Perelman School of Medicine at the University of Pennsylvania. Their findings, published today in Cell Reports, pave the way for a clinical trial to test the addition of lidocaine to the standard of care therapy for patients with head and neck cancers. The local anesthetic drug has long been suggested to have beneficial effects in cancer patients, but it wasn’t known how or why.

The preclinical study was led by Robert Lee, PhD, and Ryan Carey, MD, both assistant professors of Otorhinolaryngology – Head & Neck Surgery, and Zoey Miller, a Pharmacology graduate student at Penn and member of Lee’s lab. The team found that lidocaine activates the bitter taste receptor T2R14, which is elevated in various cancer cells. When this receptor is activated, it starts a process called apoptosis, causing the cancer cells to die. The specific mechanisms that allow lidocaine to activate T2R14 are mitochondrial calcium ion overload, which produces reactive oxygen species that can damage biomolecules, and proteasome inhibition, together resulting in cell death.
Previous work by the team showed that bitter taste receptors are found in many oral and throat cancer cells, where they trigger apoptosis, and that increased expression of these bitter receptors is correlated with improved survival outcomes in patients with head and neck cancer. In April 2023, a multi-institutional randomized clinical trial published in the Journal of Clinical Oncology found that breast cancer survival increased when lidocaine was administered before surgery.
“We’ve been following this line of research for years but were surprised to find that lidocaine targets the one receptor that happened to be most highly expressed across cancers,” Lee said. “T2R14 is found in cells throughout the body. What’s incredibly exciting is that a lot of existing drugs that activate it, so there could be additional opportunities to think about repurposing other drugs that could safely target this receptor.”
While T2R14 helps the body perceive bitter taste in the mouth, the function of the receptor in other cells throughout the body is unclear. Lidocaine is typically injected into the skin or other tissues to prevent pain by blocking nerve signals and could easily be directly injected near or around accessible oral tumors.
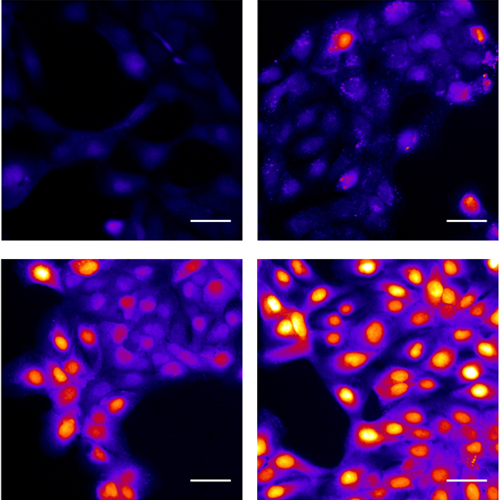
“Speaking as a head and neck surgeon, we use lidocaine all the time,” Carey said. “We know lidocaine is safe, we’re comfortable using it, and it’s readily available, which means it could be incorporated into other aspects of head and neck cancer care fairly seamlessly.”
The study, which was done primarily in cell lines of head and neck squamous cell carcinomas (HNSCCs), also found that T2R14 is particularly elevated in HNSCCs associated with the human papillomavirus (HPV), which is now the dominant form of HNSCC. As a result of these findings, Carey is planning to develop a clinical trial at Penn Medicine’s Abramson Cancer Center to test the addition of lidocaine to standard care for HPV-associated HNSCCs.
“While we’re not suggesting the lidocaine could cure cancer, we’re galvanized by the possibility that it could get an edge on head and neck cancer treatment and move the dial forward, in terms of improving treatment options for patients with this challenging form of cancer,” Carey said.
The study was supported by the National Institutes of Health (T32GM008076, R01DC016309, R01AI167971, and R21DC020041), the Blavatnik Family Foundation, the American Head and Neck Society, and the McCabe Foundation.
To learn more about clinical trials at Abramson Cancer Center and eligibility to participate click here or call 1-855-216-0098 to speak to a clinical trial navigator.
